Lipid bilayers emerge by self-organization of amphiphilic molecules and are the essential component of membranes of living cells. An important task of them is the selective exchange of substances between the cell and its environment. This becomes particularly interesting for delivering foreign molecules and RNA into the cell. In the classical view of cell biology static structures such as pores and channels formed by specific proteins control the translocation of molecules. In this work we show that there exists a straight forward mechanism for translocation of polymers through lipid bilayers if the monomers of the chain show a certain balance of hydrophobic and hydrophilic strength. Using the bond fluctuation method with explicit solvent to simulate the self-organized lipid bilayer and the polymer chain we show that the chain is adsorbed by the bilayer at a critical hydrophobicity of the monomers. At this point all monomers have an intermediate degree of hydrophobicity which is large enough to overcome the insertion barrier of the ordered lipids, but still small enough to avoid trapping in the core of the bilayer. In a narrow range around this critical hydrophobicity the chain can almost freely penetrate through the model membrane whose hydrophobic core becomes energetically transparent here.
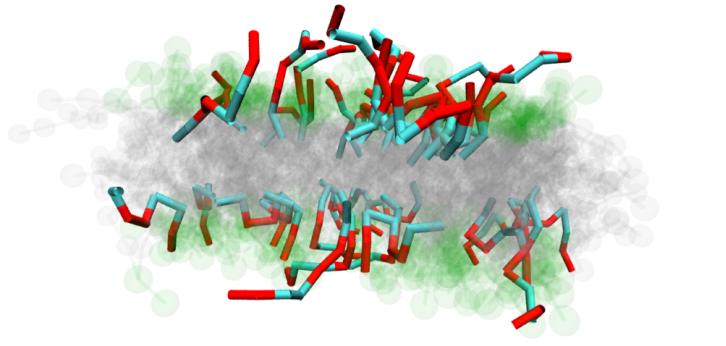
Our simulations also allow to calculate the permeability of the membrane with respect to the solvent. We show that the permeability is strongly increased close to the critical hydrophobicity suggesting that here the perturbation of the membrane patch around the adsorbed chain is highest.
name=”Jens-Uwe Sommer”
photo=”https://softmat.net/wp-content/uploads/2012/05/sommer1.jpg”
url=”https://softmat.net/t2t_products/sommer/”
] [people_short
name=”Vladimir Baulin”
photo=”https://softmat.net/wp-content/uploads/2012/05/baulin1.jpg”
url=”https://softmat.net/t2t_products/baulin/”
]

Suresh Ki
I am a polymer chemist, interested to collaborate in the design and development of hydrophobic-hydrophilic block copolymers